What We Know (& A Few Thing We Don’t) About COVID-19 Vaccines: A Conversation with Dr. Jeff Duchin
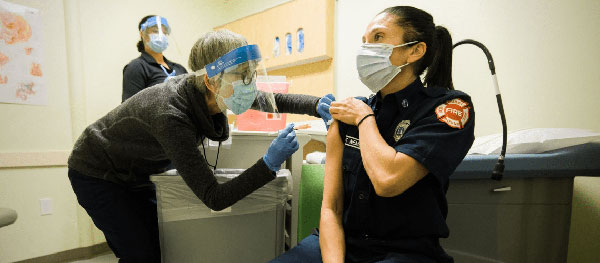
Two COVID-19 vaccines have been authorized for emergency use by the FDA and the first shipments have arrived in King County. We checked in with Dr. Jeff Duchin, Health Officer for Public Health—Seattle & King County, to learn more about this major development in the response to the pandemic.
It’s exciting that the first vaccinations have already been given in King County. Who is getting the first shots?
Initially the vaccine will be in very short supply, so they’ll be going to frontline healthcare workers who have been taking care of us throughout this pandemic—health care workers in hospitals, clinics, and other settings who are at high risk of being exposed to COVID-19 and to healthcare workers and residents in long-term care facilities. Over time, as more doses become available, they’ll be offered to more people.
How effective are the new vaccines?
These vaccines hit the ball out of the ballpark with respect to effectiveness. In the initial large clinical trials, the Pfizer and Moderna vaccines are preventing about 94%–95% of infections, which is phenomenal and comparable to our most effective vaccines. Time will tell how long protection lasts and whether booster shots will be needed down the road.
It’s great that these vaccines are highly effective. What about the safety? Some people have concerns because they were able to produce the vaccines so quickly.
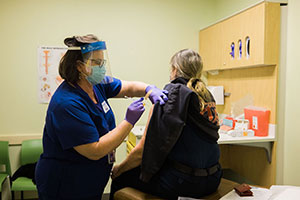
Public Health—Seattle & King County staff administering COVID-19 vaccine to a local emergency responder.
These vaccines were developed much faster than usual, so it’s understandable that people would wonder about safety. That’s why it’s important to know that in the making of these vaccines, there were no safety steps skipped in the clinical trials or evaluation of data for safety. Usually, required stages in vaccine development and testing are done in time-consuming, separate steps over several years, one after the next. Because of the pandemic, the federal government provided special funding to vaccine researchers and manufacturers to allow testing and production to happen at the same time. This ensured that if a vaccine was found to be safe and effective, there would already be a supply to roll out.
The data for these vaccines was evaluated with scientific rigor and independent review to ensure it met FDA’s standards for safety, effectiveness, and quality. So, the process was made quicker but still thorough and focused on ensuring safety and effectiveness.
These vaccines received an Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA). How is that different from the usual vaccine approval process?
The Food and Drug Administration (FDA) uses Emergency Use Authorizations, known as EUAs, during national emergencies to make a product available to address an urgent health need. The FDA has issued EUAs to address anthrax, Ebola, H1N1 and other health emergencies in the past. The EUA allows a product to be used before it has a full license. When the clinical trials are completed, the vaccine manufacturers are expected to apply for the usual licensure application from FDA. This review process must address statutory and regulatory requirements.
You can find out more about when EUA are used and the process for authorization from the FDA website.
Who was included in the vaccine studies and was the vaccine effective across groups?
The Pfizer vaccine clinical trials included over 40,000 people aged 16 years and older in the U.S., Argentina, Brazil, and South Africa. About 30,000 Americans aged 18 years and older volunteered for the clinical trials for the Moderna vaccine.
- Ten percent of the volunteers for both vaccines were Black or African American, about 4%–5% were Asian, over 20% were Hispanic/Latino, and 0.5%–1% were American Indian/Alaskan Native.
- Twenty percent of the Pfizer volunteers and 17% of the Moderna volunteers had an underlying health condition such as diabetes, obesity, or lung disease.
The vaccines were shown to be effective across these groups.
The two vaccines that have been authorized from Pfizer and Moderna are mRNA vaccines. What are mRNA vaccines and how do they work?
It’s an amazing technology that has been in the works for decades. mRNA is basically a molecular blueprint that tells your cells’ natural protein-making machinery to make a protein that resembles an important protein on the COVID-19 virus. When your cells make that protein, your body creates a very strong immune response and antibodies against it that protects against COVID-19. So, your body learns to recognize the presence of the coronavirus and protect against it without exposure to the coronavirus.
There’s no virus in the mRNA vaccine, so you cannot get an infection from this vaccine. The mRNA is broken down and eliminated from the body quickly after it does its work. Vaccine mRNA does not get into the nucleus of the cell or into the DNA.
What kind of common side effects do the vaccines have?
It’s common for people to have temporary symptoms like headache, a sore arm, fatigue, muscle aches, or fever for a few days after getting the shot. These side effects are more common after the second dose of vaccine. These are all signs that the body’s immune system is building up protection. This is normal and can also be seen after other commonly used vaccines, like the one for shingles.
What about the severe allergic reactions that the news has reported in a handful of people?
A small number of cases of severe allergic reactions have been reported among several hundred thousand people that have been vaccinated in the US. These reactions, called anaphylaxis, can be life threatening but are readily treatable. They can also occur with other vaccines and medications that are given to large numbers of people. That’s why there are protocols in place to monitor for safety after the vaccine is administered.
It’s too early to know the exact reason for the reactions or what ingredients in the vaccine, if any, may have triggered the reactions. This type of reaction is very rare, but as a precaution, anyone with a history of having an anaphylactic reaction to any ingredient in the COVD-19 vaccine should not get the vaccine at this time. An anaphylactic reaction would occur soon after getting the vaccination, so as a precaution, people will be asked to wait at the vaccination site for a short time after getting vaccinated and vaccinators will be prepared with epinephrine and trained to respond in case of a severe reaction.
With vaccine arriving, will we still need to wear masks and practice social distancing?
Yes, for the time being. The vaccine will give a high level of protection against infection for the person who is vaccinated, although not 100%. Also, we don’t yet know how well the vaccine prevents people from spreading the virus. That means it’s possible that someone who is vaccinated may get infected but not get sick yet still be able to spread COVID-19 to others. Studies are in progress to answer this question. And because it will take months for enough people to be vaccinated and protected, we’re all going to need to practice the key COVID-19 prevention steps for now, including wearing masks, limiting indoor activities outside of the home, avoiding crowded indoor spaces, keeping contacts with others brief and distanced, improving ventilation indoors, and washing hands.
I know everyone is exhausted from this tough year, but to help keep our loved ones and communities safe we need to hang in there for just a few more months with these tough but effective measures.
How are you feeling about this vaccine as we head into the new year?
I feel hope. This vaccine doesn’t mean the immediate end of the pandemic, but I feel it represents the beginning of the end. But in order for the vaccine to work, people have to get it. We know that not everyone is ready to get vaccinated and may have valid reasons to feel uncertain about it. I hope people will take the time to get the information that will help them make the best decision for themselves and their communities.
For more information about the COVID-19 vaccine, go to our COVID-19 vaccine website.
Contributor Meredith Li-Vollmer is a Risk Communication Specialist with Public Health—Seattle & King County.
Originally posted in Public Health Insider on December 23, 2020.
![AgeWise King County [logo]](https://www.agewisekingcounty.org/wp-content/themes/agewisekingcounty/images/logo.png)